Leeds Teaching Hospitals NHS Trust is testing AI for prostate cancer diagnosis.
- The trust collaborates with Lucida Medical for this groundbreaking trial.
- AI tool aims to streamline the diagnostic pathway, reducing waiting times.
- Prostate cancer is prevalent among men, with distinct risks for black males.
- The trial will compare AI results against outcomes from 100 patients.
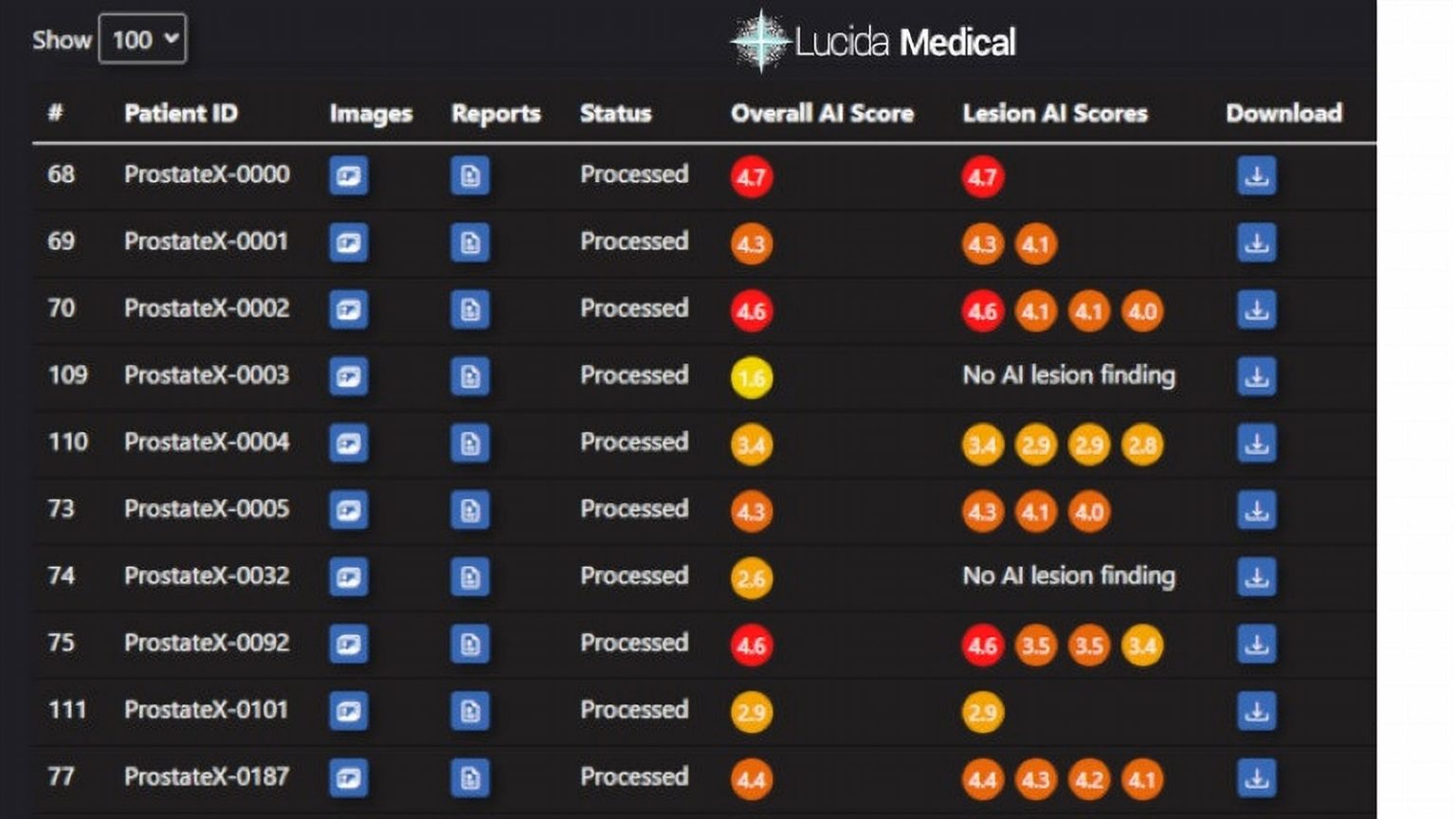
Leeds Teaching Hospitals NHS Trust (LTHT) has embarked on a significant pilot study, implementing a novel artificial intelligence tool to enhance the diagnostic process for prostate cancer. Conducted in partnership with Lucida Medical, this initiative seeks to evaluate the efficiency of the ‘Prostate Intelligence’ AI system in identifying cancerous lesions on MRI scans. Prostate cancer continues to be the most frequently diagnosed cancer among men, with statistics indicating that one in eight men in the UK will face this diagnosis in their lifetime. For black men, this risk increases to a striking one in four.
The diagnostic pathway for prostate cancer is notably intricate, and LTHT aims to leverage this AI technology to expedite the procedure, particularly in prioritising patients with cancer. By automating certain elements of the diagnostic process, the AI tool presents the potential to enhance operational efficiency, potentially allowing radiologists to manage and report on a larger number of cases daily.
The AI software under trial has been designed to analyse MRI scans with remarkable precision, assessing risk scores and prostate dimensions, which are critical in determining biopsy and subsequent treatment decisions. It functions by employing advanced algorithms to detect indicative patterns or “signatures” of cancer based on extensive datasets, including MRI and biopsy data from patients in both the Netherlands and the UK.
Consultant radiologist Dr Oliver Hulson underlined the objective of the study: “Our goal is to determine if this AI tool can accurately identify prostate cancer without underestimating or overestimating the likelihood based on MRI scans. If successful, this could enable us to streamline our prostate cancer pathway and reduce the waiting time for a biopsy from the current two to three weeks to less than one week.” Faster diagnostics are anticipated to lead to swifter treatments and improved patient outcomes.
The current study compares AI-generated results with real-world outcomes for 100 patients who have recently completed their prostate cancer diagnostic journey. The research anticipates that the AI tool will offer a sensitive and specific method for detecting prostate cancer, possibly revolutionising the diagnostic pathway.
As proposed, the AI integration could ultimately enable a ‘one-stop’ diagnostic approach wherein patients can undergo an MRI scan, receive a comprehensive review, and if indicated, proceed to biopsy all within a single day. This condensed timeline starkly contrasts the existing elongated waiting periods and has the potential to significantly diminish patient anxiety associated with prolonged diagnostic delays. Dr Hulson expressed hope that “such advancements would provide patients with their results at the earliest possible opportunity,” thereby improving the overall standard of care.
The trial at LTHT signifies a potential turning point in prostate cancer diagnostics, aiming to facilitate faster, more accurate patient care.